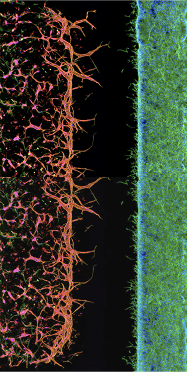
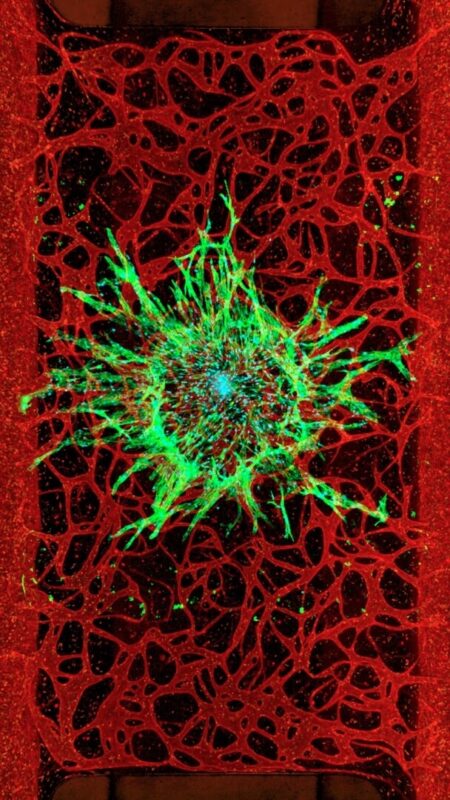
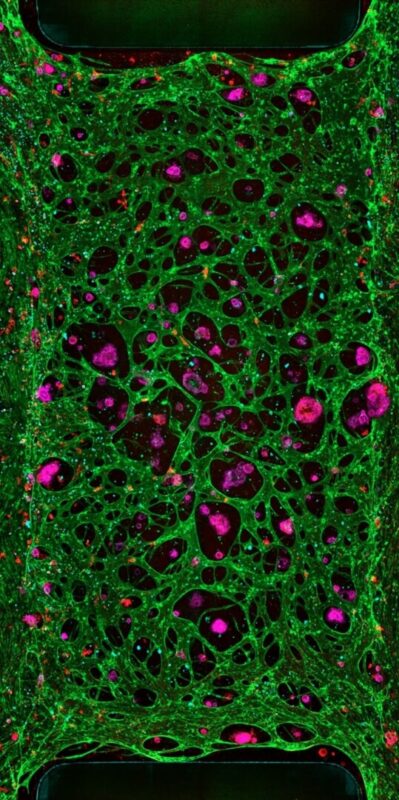
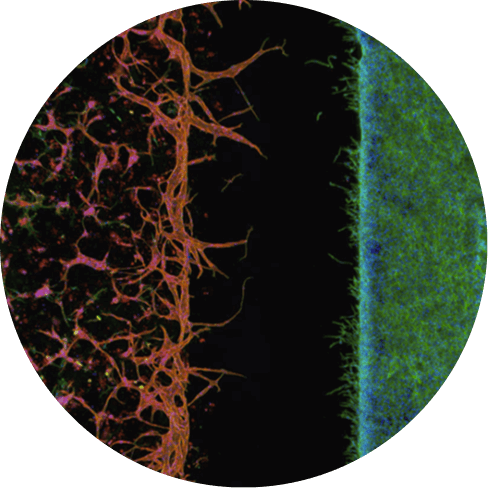
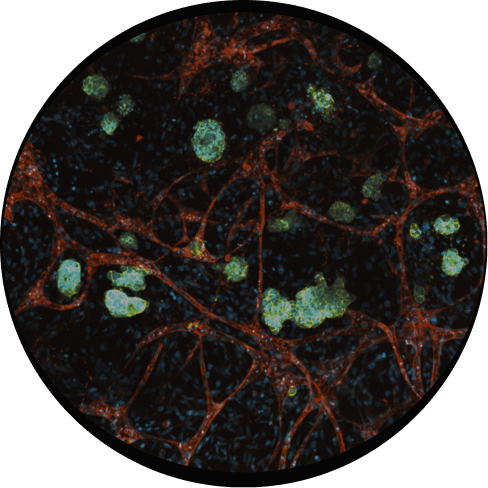
Human Cells
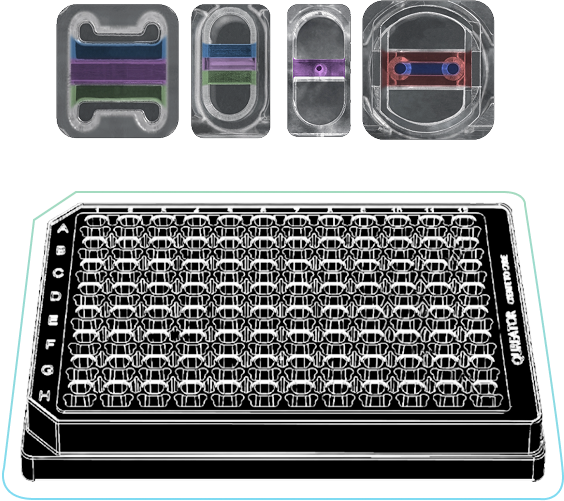
CurioChips
Patient-derived Data
Omics & Literature
Databases
Qureator Human Disease Models
Testing Modalities:
- Small molecule
- Biologics/Antibodies
- Cell & Genen Therapies
- CRISPRi
- etc…
New Target Identification
Discovery of Synergistic Combinations
Efficacy & Dosage Prediction
High Throughput MPS and AI
and Omics
Extraction
Analysis
Best Drug Candidates
Patient Stratification
Qureator’s Historic FDA IND Approval Based on Human-Relevant Data Highlighted in The BIO
Qureator extends its sincere appreciation to The BIO for its in-depth reporting on our landmark FDA IND approval achieved without animal PoC data. The coverage underscores a transformative moment for the global drug-development landscape and highlights the pivotal collaboration between Qureator and SillaJen.
Paul Park Joins Qureator as Chief Business Officer
Qureator Appoints Paul Park as Consulting Chief Business Officer
We are thrilled to welcome Paul Park to Qureator as our new Consulting CBO. Paul brings over 20 years of executive experience from Amgen, Ionis, and Circle Pharma, with a proven track record in strategy, M&A, and high-impact partnerships.
Qureator Shortlisted for Prestigious Lush Prize in Science
Qureator is proud to announce its selection as a shortlisted finalist for the renowned Lush Prize in the Science category. This prestigious initiative awards £50,000 to projects that drive the transition toward practical, regulator-accepted non-animal testing methods.
View the full shortlist: https://lushprize.org/2026-prize/2026-prize-shortlist/